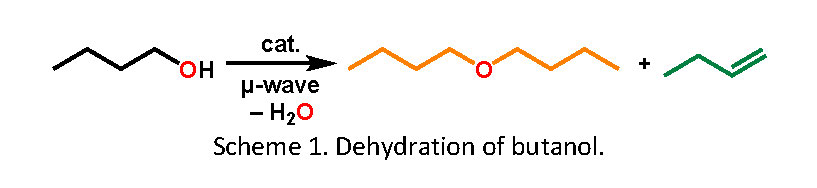
Today, implementing the use of sustainable feedstocks is a vital driver across the chemicals’ manufacturing industries. The dehydration of bio-derived butanol to the corresponding n-butene and/or the di-n-butyl ether has been attracting great industrial interests for decades. Di-n-butyl ether (DnBE) has a variety of uses, for example, DnBE is a safer substitute to tetrahydrofuran (THF) for organometallic chemistry and for Grignard reactions.1 The other product, n-butene, can be further converted (via metathesis) to propene, for which demand remains high for use in various applications, for example, the synthesis of polypropylene.
Herein, we will present our studies around the selective, catalytic dehydration of butanol to n-butene and/or di-n-butyl ether using different methods including under microwave irradiation. Microwave reactor has more energy efficient heating compare to the conventional process. The advantage of heterogeneous catalysis in microwave is that the solid catalyst absorbs microwave irradiation more significantly and can be considered as an internal heating source.2 To the best of our knowledge, there are no reported examples on butanol dehydration under microwave irradiation mediated by a heterogeneous catalyst. We will discuss the importance of using the microwave irradiation and optimised conditions on the dehydration of n-butanol to the corresponding ethers. During this study a range of catalysts have been screened, zeolites (HZSM-5 and H-beta) tends to give the best results. The selectivity of di-n-butyl ether could be optimised by varying the Si: Al ratio of zeolites. With the best results, over 90% conversion and over 90% selectivity of the di-n-butyl ether has been achieved after the butanol dehydration reaction, with mainly the gaseous butene as the other product.
- J. E. Baldwin, E. J. O’Neil, J. Label. Compd. Radiopharm, 2009, 52, 427
- V. Palma, D. Barba, M. Cortese, M. Martino, Catalysts, 2020, 10, 246
Yiping Shi (Durham), Phil Dyer (Durham), Andy Weller (York)
Watch the talk below:
Biography

Dr. Yiping Shi (Sherry) comes from China and completed the MSci degree in Chemistry with one year industrial placement in the University of Bristol. She then completed her PhD (2018) in Chemistry in the University of St. Andrews, working under the supervision of Prof. Paul Kamer and Prof. David Cole-Hamilton. Her PhD research covers organic synthesis, homogeneous catalysis and organometallic chemistry, and was focused on the development of new catalytic routes for the production of value-added materials, such as polymer precursors and pharmaceutical drugs from waste biomass (eg. tall oil and cashew nut shell liquid). After her PhD, she then worked on two industrial projects under the supervision of Dr. Paul Webb in the same department, concentrated on the heterogeneous catalysis to convert food waste into value-added materials using mainly flow systems. On the 1st of July she became a postdoctoral research associate in the University of Durham, working under the supervision of Dr. Phil Dyer (Durham) and Prof. Andrew Weller (Oxford) on a project entitled “Cascade catalysis: bio-derived butanol to propene via dehydration and metathesis.”




