Homogeneous/biocatalysis in traditional aqueous media often suffer from poor substrate solubility, and difficult catalyst/product separation and subsequent catalyst recycle. Ionic liquids possess a number of tunable properties favorable for supporting homogeneous/biocatalysis integrity by adjusting ionic liquids hydrophobicity, hydrophilicity, acid-base properties and intermolecular interactions, hence, ionic liquids are emerging as a reaction medium for both homogeneous and biocatalytic reactions.1,2,3,4 Catalyst immobilization is one of the expanding research areas, which can in principle efficiently overcome the many challenges in catalytic processes. We are addressing challenges in aqueous biocatalytic and homogeneous catalytic processes for green chemistry and sustainable chemical production in water. In our research, we are developing new materials and methods for homogeneous/biocatalyst immobilization that would significantly contribute towards sustainable homogeneous/biocatalyst process development. Currently, we have successfully entrapped homogeneous and biocatalysts in silica and soft gels of a range of ionic liquids (Fig 1).
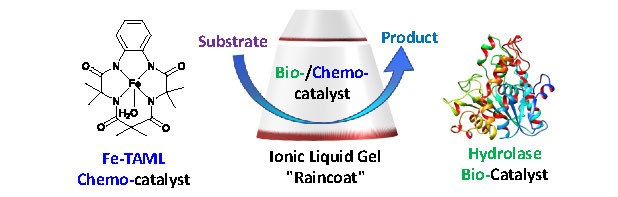
Homogeneous transition metal (Fe) catalysts5 and the biocatalysts were entrapped in ionic liquid gels. The constituents of the immobilized gel systems were characterized by UV-Visible, NMR, FTIR spectroscopy, Mass spectrometry and elemental analysis. The hydrolytic/degradation catalytic activity of free and immobilized homogeneous/biocatalysts were measured by UV-Visible spectroscopy. We have demonstrated that immobilized homogeneous/biocatalysts are active within ionic liquid gels.
Acknowledgement
This work is supported by Engineering and Physical Sciences Research Council (UK) grants EP/ R026645/1, The UK Catalysis Hub Science: 2 Catalysis at the Water-Energy Nexus, and EP/M013219/1, Biocatalysis & Biotransformation: A 5th Theme for the National Catalysis Hub. Thanks to our collaborator Professor Terrence J. Collins (Carnegie Mellon University, USA) and Dr Stefan Mix, Almac Bioscience, Belfast, Northern Ireland, UK.
Hasan T. Imama, Patricia C. Marr*a and Andrew C. Marr*a
aSchool of Chemistry and Chemical Engineering, Queen’s University Belfast, Belfast, UK,
*Corresponding authors: p.marr@qub.ac.uk, a.marr@qub.ac.uk
References
- K. M. Bothwell, V. Krasňan, F. Lorenzini, M. Rebroš, A. C. Marr and P. C. Marr, ACS Sustain. Chem. Eng., 2019, 7, 9948–9956.
- P. C. Marr and A. C. Marr, Green Chem., 2016, 18, 105–128.
- M. Moniruzzaman, K. Nakashima, N. Kamiya and M. Goto, Biochem. Eng. J., 2010, 48, 295–314.
- H. Zhao, J. Chem. Technol. Biotechnol., 2010, 85, 891–907.
- P. McNeice, A. Reid, H. T. Imam, C. Mcdonagh, J. D. Walby, T. J. Collins, A. C. Marr and P. C. Marr, Environ. Sci. Technol., 2020, 54, 14026−14035.

Dr Hasan Tanvir Imam obtained his undergraduate and master’s degree in chemistry from Shahjalal University of Science and Technology, Bangladesh. He was awarded a Warwick University Chancellor scholarship for his PhD (2011-2015) to work with Professor Claudia Blindauer and carried out his research on understanding the metal-cluster and metal binding dynamics of two cysteine rich metalloproteins homologs from plant Arabidopsis thaliana. After completing his PhD, he spent one-year (2016-2017) in Texas A&M University, USA as a visiting research associate working on the biophysical basis of Protein Kinase Cα-Peptidylprolyl isomerase (Pin1) binding and catalysis with Dr Tatyana Igumenova. He returned to the UK (2017) and joined the University of St-Andrews as a postdoctoral research fellow in a Catalysis Hub funded collaborative research project on artificial metalloenzymes for hydroformylation, working with Professor Paul Kamer, Dr Andrew Marr, and Dr Amanda Jarvis. This artificial metalloenzymes catalytic work inspired him to continue his research on artificial metalloenzymes and biocatalysis (2019-present) with Dr Andrew Marr and Dr Patricia Marr in Queen’s University Belfast in another UK Catalysis Hub funded project. Currently, he is developing strategies to stabilize chemo-and biocatalysts in ionic liquid based soft gel biomaterials for green and sustainable chemical transformations in aqueous and aqueous biphasic media.
View this talk below:




