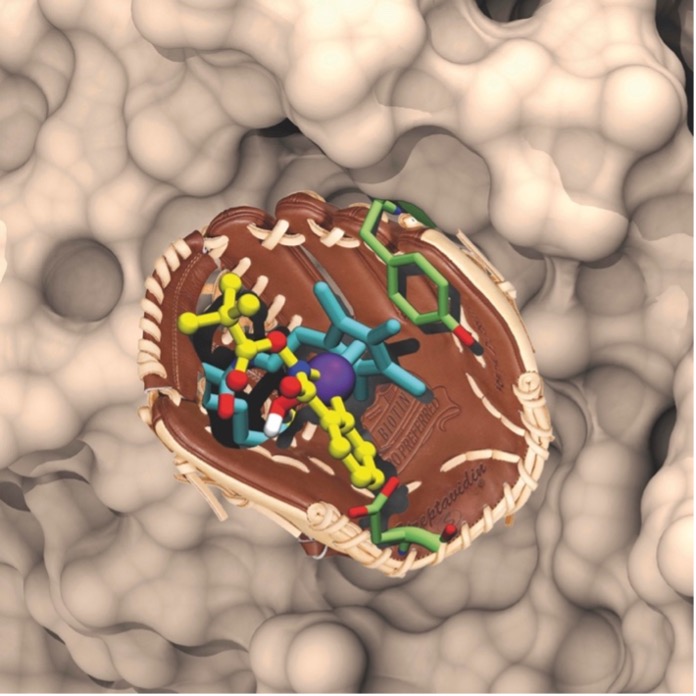
Artificial metalloenzymes (ArMs) result from the incorporation of an abiotic metal cofactor within a host protein, Figure 1. Thanks to the remarkable supramolecular affinity of biotin for streptavidin (KD > 10-13 M), linking a biotin anchor to an organometallic catalyst ensures that, upon addition of streptavidin, the abiotic cofactor is quantitatively incorporated within the streptavidin. Alternatively, human carbonic anhydrase has proven equally versatile for the development of artificial metalloenzymes relying on aryl-sulfonamide anchors to ensure the localization of the abiotic metallocofactors within the host protein. The catalytic performance of the resulting ArMs can be optimized either by chemical (variation of the anchor-spacer-ligand moiety) or genetic- (mutation of the host protein) means. These chemo-genetic schemes were applied to optimize the performance for thirteen different catalyzed transformations as well multiple reaction cascades in the presence of natural enzymes, either in vitro or in vivo.
This presentation will highlight the group’s effort to implement and evolve artificial metalloenzymes with activities that are complementary to natural enzymes. These include: C–H activation, hydroxylation, olefin metathesis, imine reduction, cross-coupling reactions etc.
Thomas R. Ward
NCCR Molecular Systems Engineering and Department of Chemistry, University of Basel, Basel, Switzerland
Email: thomas.ward@unibas.ch,
Web: https://ward.chemie.unibas.ch/en/home/
Biography

Thomas Ward received his diploma in Chemistry in 1987 from the University of Fribourg and his PhD from the ETH Zürich. He then joined the group of Prof. R. Hoffmann at Cornell University as a postdoctoral fellow to work on qualitative MO theory. After a second postdoc at the University of Lausanne, he moved to Berne to undertake his independent career in 1993. In 2000, he moved to University of Neuchâtel and in 2008 to the University of Basel. His research focuses on artificial metalloenzymes and bio-nanotechnology.
Thomas Ward was awarded the ETH Medaille (1991), the A. Werner Fellowship (1993) as well as the A. Werner Prize (1998), a Swiss National Science Foundation Professorship (2000) and the RSC award in BioInorganic Chemistry (2017). He was awarded an advanced ERC grant to realize his DrEAM: the Directed Evolution of Artificial Metalloenzymes.




